Citric Acid Mediates the Iron Absorption from Low Molecular Weight Human Milk Fractions | Journal of Agricultural and Food Chemistry

Chromium (VI) reduction in the nano- or micron-sized iron oxide – Citric acid systems: Kinetics and mechanisms - ScienceDirect

In-Situ Preparation and Characterization of Aconitic Acid Capped Fe3O4 Nanoparticle by Using Citric Acid as a Reducing Agent | SpringerLink
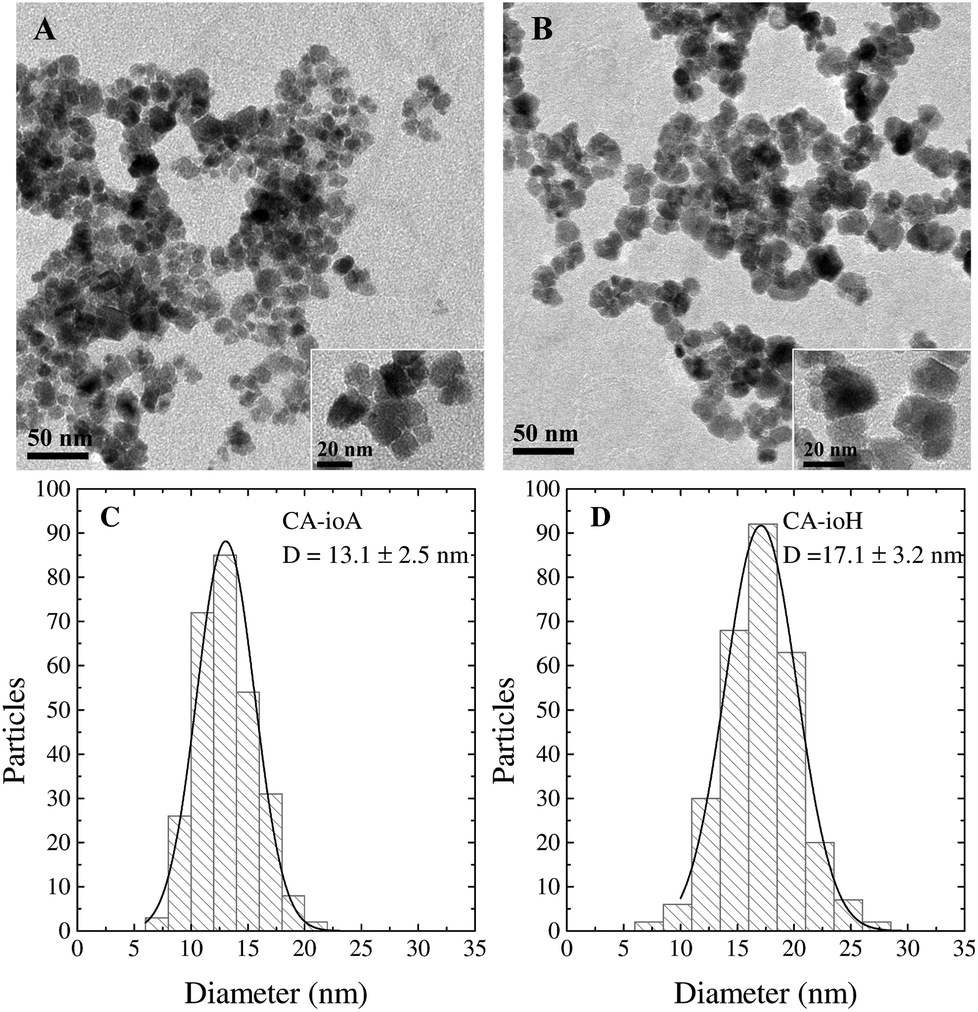
High performance multi-core iron oxide nanoparticles for magnetic hyperthermia: microwave synthesis, and the role of core-to-core interactions - Nanoscale (RSC Publishing) DOI:10.1039/C4NR06239F
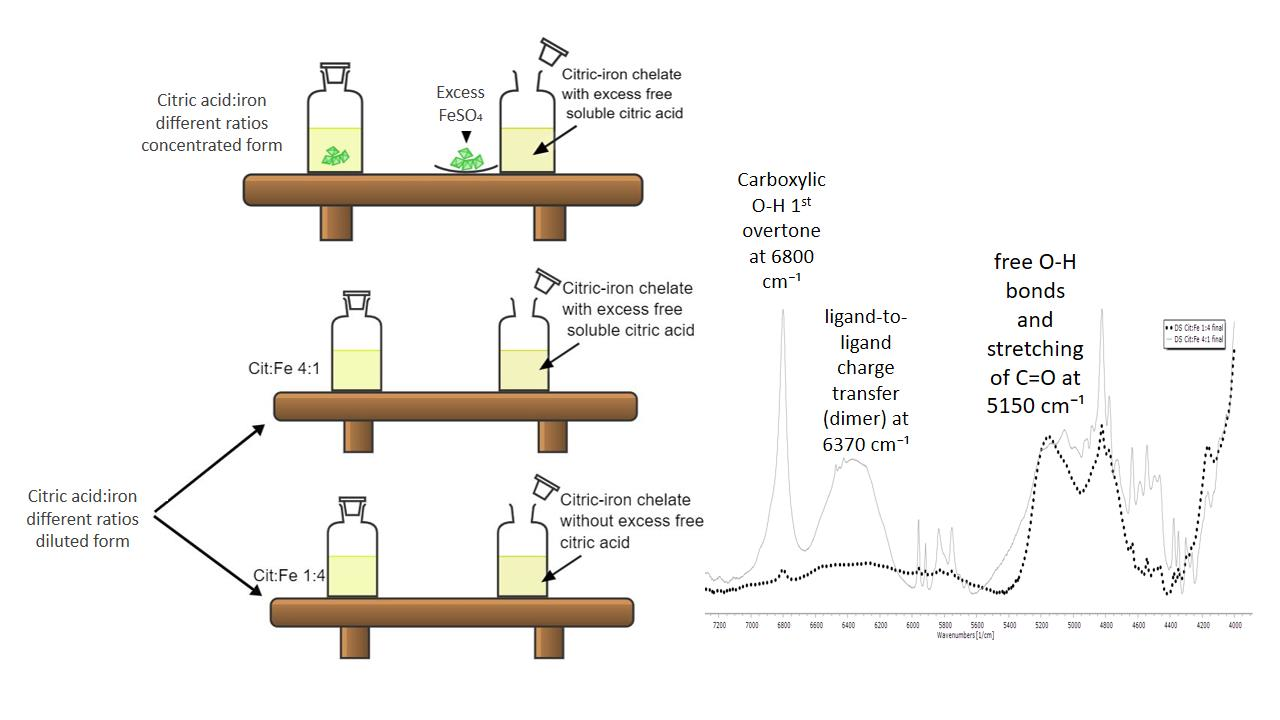
Foods | Free Full-Text | Are Citric Acid-Iron II Complexes True Chelates or Just Physical Mixtures and How to Prove This?

Effect of synthesis conditions on the properties of citric-acid coated iron oxide nanoparticles - ScienceDirect

a) Stable aqueous dispersion containing citric acid-and PAAcoated Fe 3... | Download Scientific Diagram
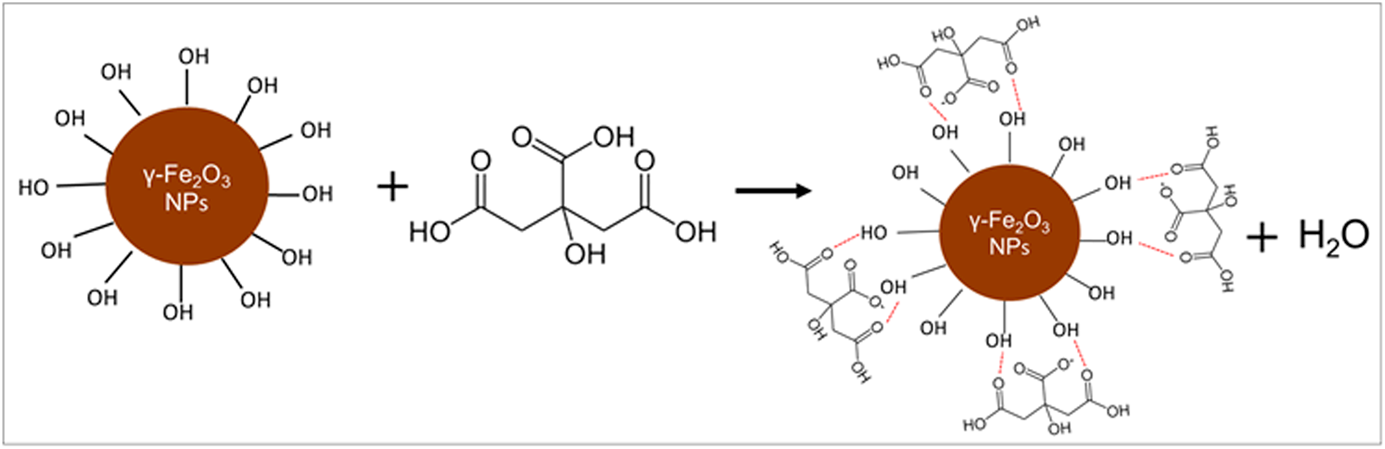
Glucose-sensing properties of citrate-functionalized maghemite nanoparticle–modified indium tin oxide electrodes | Journal of Materials Research | Cambridge Core
Effect of ascorbic acid and citric acid on bioavailability of iron from Tegillarca granosa via an in vitro digestion/Caco-2 cell culture system - Food & Function (RSC Publishing)

Enhancing the reactivity of bimetallic Bi/Fe0 by citric acid for remediation of polluted water - ScienceDirect
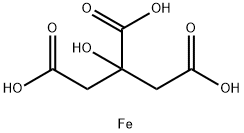
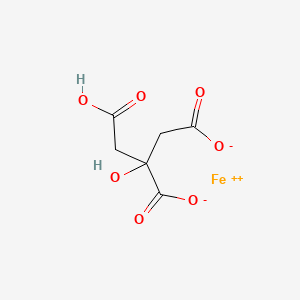
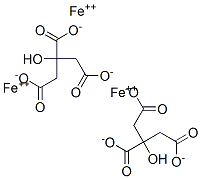
3522-50-7 | Iron(III) Citrate (Technical Grade) | 2-Hydroxy-1,2,3-propanetricarboxylic Acid, Iron(3+) Salt (1:1); Citric Acid, Iron(3+) Salt (1:1) (8CI); Fe(III)-Citrate Complex (1:1); Ferric Citrate; Zerenex | C6H8O7 • (Fe) | TRC

Degradation of carbamazepine by singlet oxygen from sulfidized nanoscale zero-valent iron – citric acid system - ScienceDirect
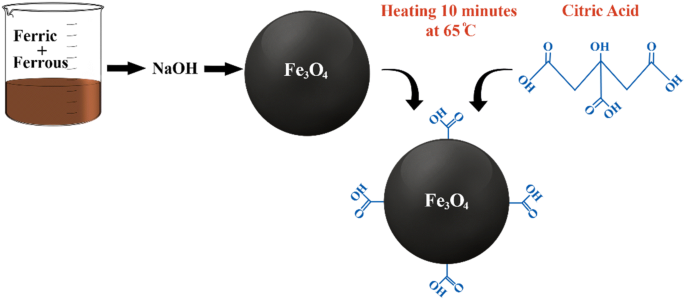
Simple rapid stabilization method through citric acid modification for magnetite nanoparticles | Scientific Reports