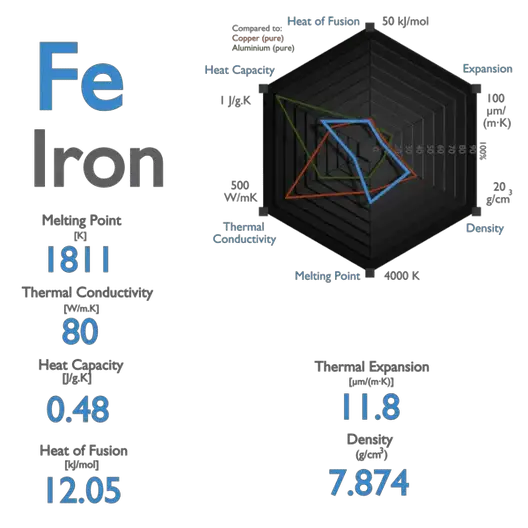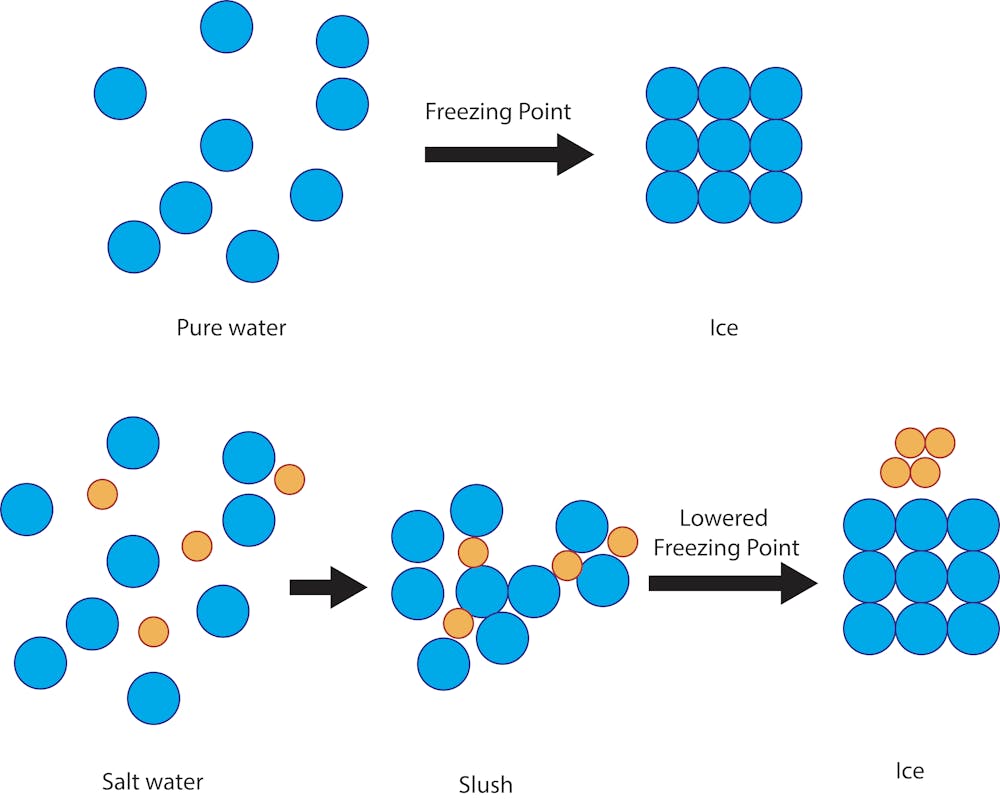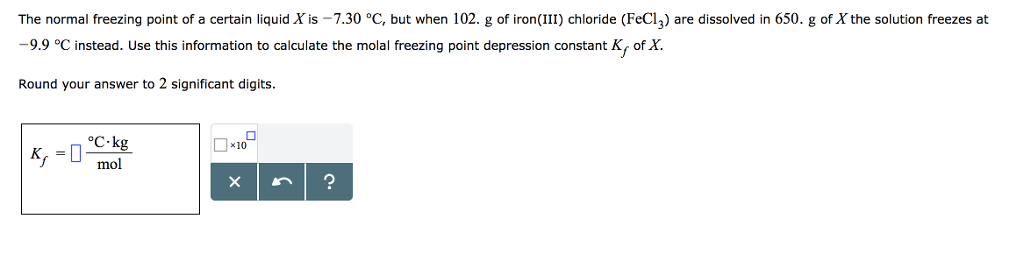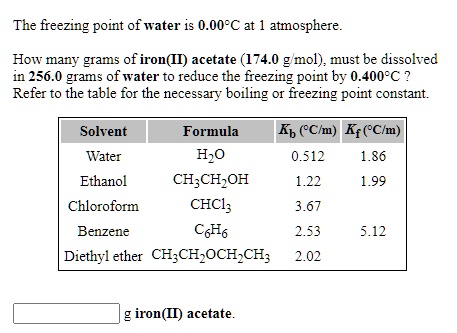
SOLVED: The freezing point of water is 0.00 C at atmosphere How many grams of iron(I) acetate (174.0 g mol): must be dissolved in 256.0 grams of water to reduce the freezing

Elements Hydrogen Number of: Protons 1 Neutrons 0 Electrons 1 Boiling point -252 Freezing point -259 State at room temperature gas. - ppt download

The Chart given below shows the boiling point and freezing point of some substances. State whether these substances are solid, liquid or gaseous at room temperature :

Freezing temperatures of aqueous iron(III) sulfate solutions and crystallization of a new acidic water-rich sulfate - ScienceDirect

a scientist has a mystery element that she wants to identify by using its properties these properties - Brainly.com
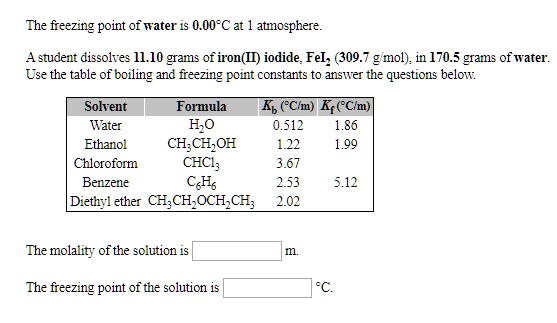
SOLVED: The freezing point of water 0.00-C at atmosphere: Astudent dissolves Il.1O grams of iron(ID) iodide; Fel; (309.7 g mol) in 170.3 grams of water: Use the table of boiling and freezing
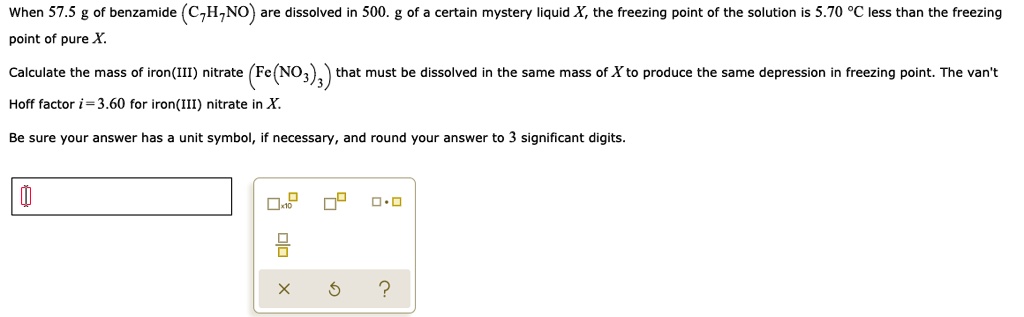
SOLVED: When 57.5 of benzamide (C,HzNO) are dissolved in 500. certain mystery liquid X, the freezing point of the solution is 5.70 %C less than the freezing point of pure Calculate the
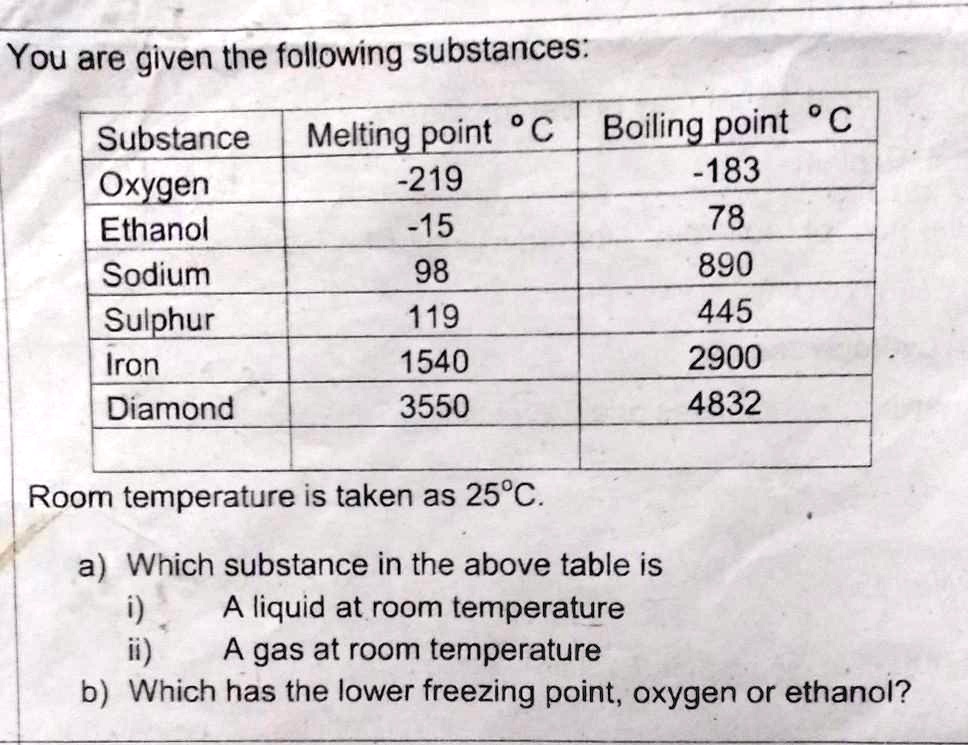
SOLVED: 'give the answer of of this question please You are given the following substances: point C Boiling point C Substance Melting -219 -183 Oxygen Ethanol -15 78 Sodium 98 890 Sulphur

Basic ice-modulation functions of CPAs. Ice freezing point depression,... | Download Scientific Diagram

Freezing temperatures of aqueous iron(III) sulfate solutions and crystallization of a new acidic water-rich sulfate - ScienceDirect




![Iron-Carbon Phase Diagram Explained [with Graphs] Iron-Carbon Phase Diagram Explained [with Graphs]](https://fractory.com/wp-content/uploads/2020/03/Iron-carbon-phase-diagram-explained.jpg.webp)