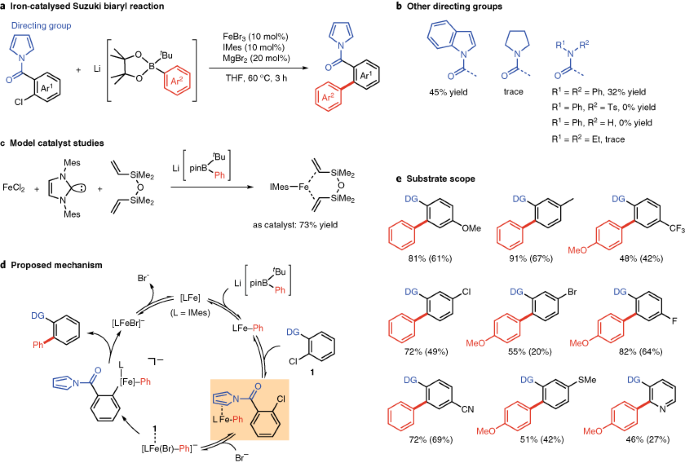
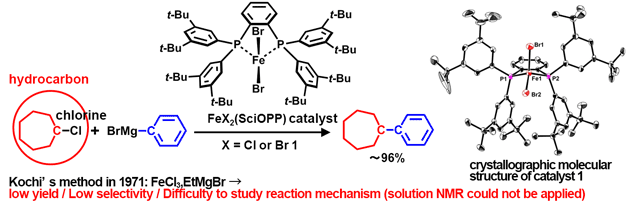
Recent advances in iron-catalysed cross coupling reactions and their mechanistic underpinning - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C5QI00295H

Iron-Catalyzed Cross-Coupling of Primary and Secondary Alkyl Halides with Aryl Grignard Reagents | Journal of the American Chemical Society

Iron-Catalyzed Suzuki–Miyaura Cross-Coupling Reactions between Alkyl Halides and Unactivated Arylboronic Esters | Organic Letters

N-Butylpyrrolidone (NBP) as a non-toxic substitute for NMP in iron-catalyzed C(sp2)–C(sp3) cross-coupling of aryl chlorides | Green Chemistry International

Iron-Catalyzed Cross-Coupling of Thioesters and Organomanganese Reagents | Organic Chemistry | ChemRxiv | Cambridge Open Engage
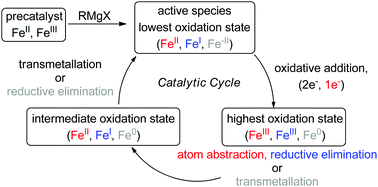
Recent advances in iron-catalysed cross coupling reactions and their mechanistic underpinning - Inorganic Chemistry Frontiers (RSC Publishing)

Iron‐Catalyzed Cross‐Couplings in the Synthesis of Pharmaceuticals: In Pursuit of Sustainability - Piontek - 2018 - Angewandte Chemie International Edition - Wiley Online Library
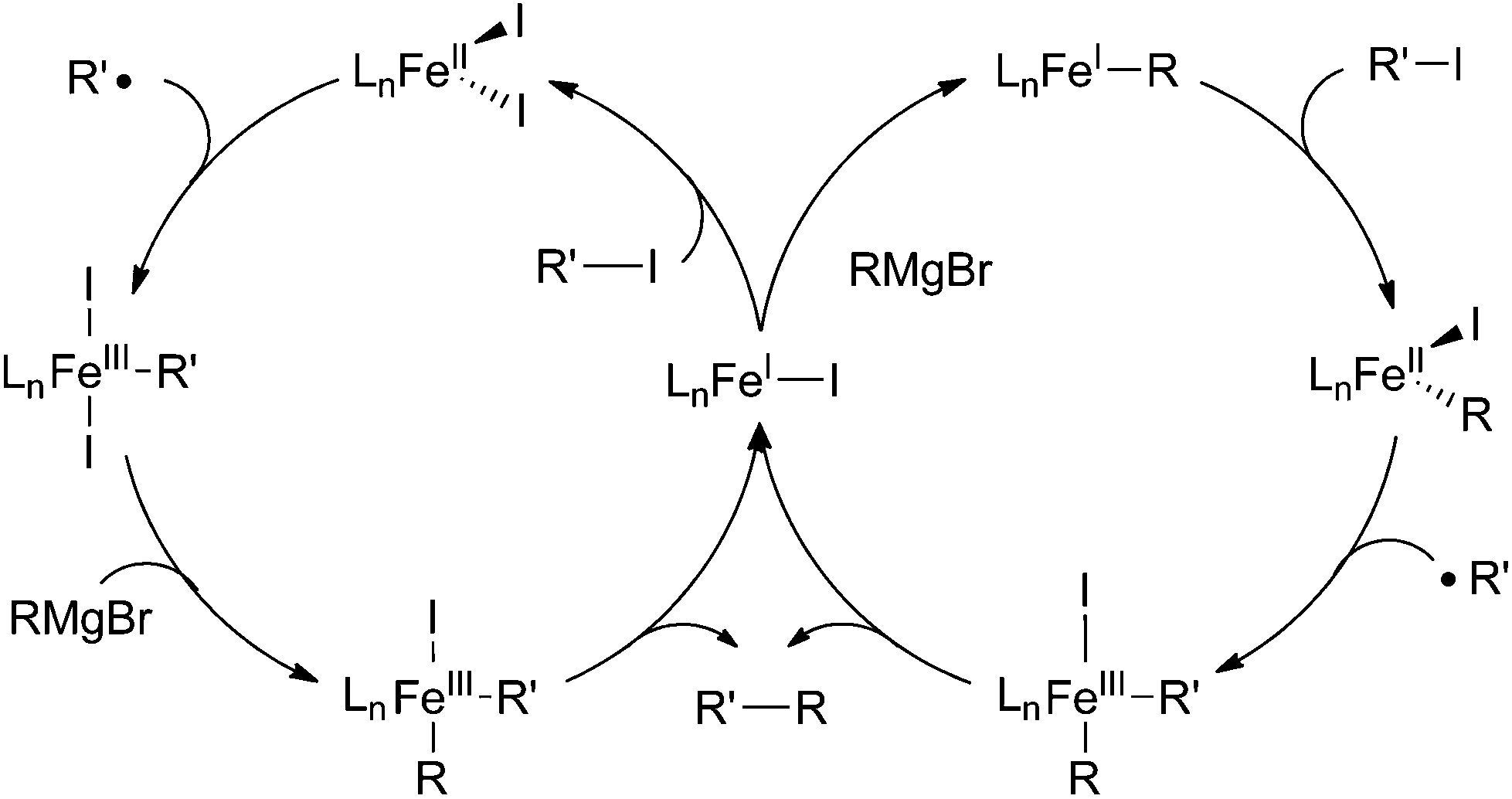
Recent advances in iron-catalysed cross coupling reactions and their mechanistic underpinning - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C5QI00295H
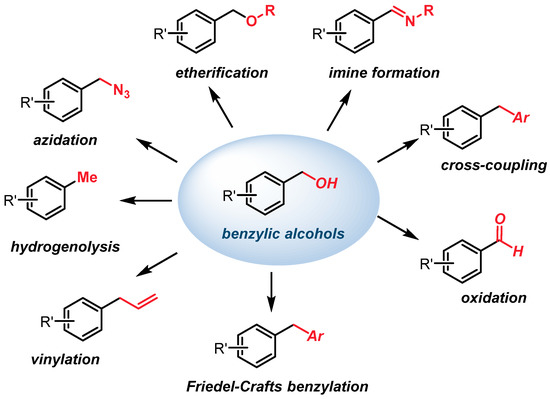
Molecules | Free Full-Text | Sequential Iron-Catalyzed C(sp2)–C(sp3) Cross-Coupling of Chlorobenzamides/Chemoselective Amide Reduction and Reductive Deuteration to Benzylic Alcohols

Recent advances in iron-catalysed cross coupling reactions and their mechanistic underpinning - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C5QI00295H

Table 2 from Iron(ii)-catalyzed sulfur directed C(sp3)-H bond amination/C-S cross coupling reaction. | Semantic Scholar

Outline of the iron-catalyzed cross-coupling reaction mechanism for the... | Download Scientific Diagram
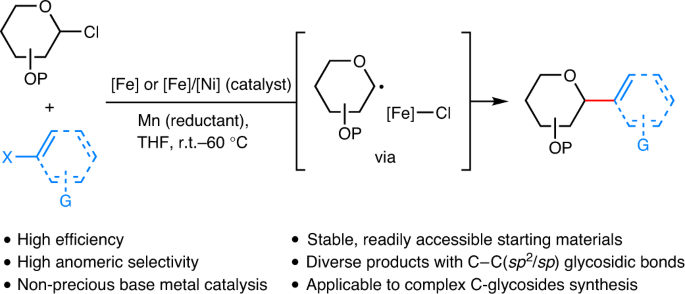
Iron-catalysed reductive cross-coupling of glycosyl radicals for the stereoselective synthesis of C-glycosides | Nature Synthesis

Iron-Catalyzed Suzuki–Miyaura Cross-Coupling Reactions between Alkyl Halides and Unactivated Arylboronic Esters | Organic Letters
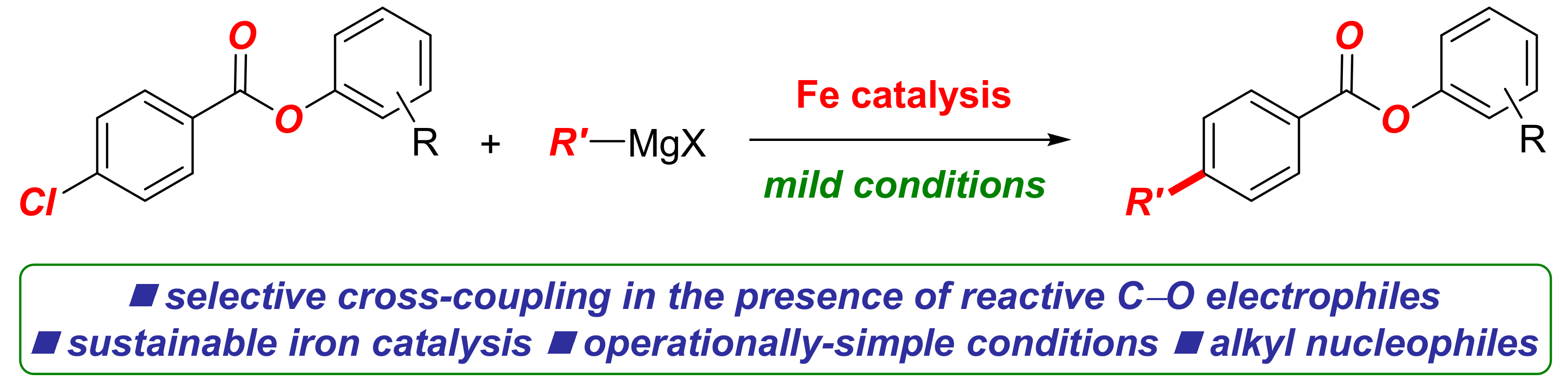
Molecules | Free Full-Text | Iron-Catalyzed C(sp2)–C(sp3) Cross-Coupling of Aryl Chlorobenzoates with Alkyl Grignard Reagents

Molecules | Free Full-Text | Iron-Catalyzed C(sp2)–C(sp3) Cross-Coupling of Aryl Chlorobenzoates with Alkyl Grignard Reagents

Recent advances in iron-catalysed cross coupling reactions and their mechanistic underpinning - Inorganic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C5QI00295H