
SOLVED: A reaction of Aluminum metal with Iron nitrate will produce Iron metal and Aluminum nitrate as shown; Fe(NOslziaql Ala) Ferl AI(NOs) (unbalanced) 1.898 of Al reacts with 68.0 mL of 1.15

SOLVED: Write the balanced molecular equation; including phases, for the reaction of aqueous sodium phosphate with aqueous iron(II) nitrate molecular equation: Na; POA(aq) + Fe(NO3)(aq) Fe;(PO4)2(s) + NaNO; (aq) Incorrect

SOLVED: A solution of Iron (II) nitrate is poured into sodium sulfide solution and an iron (II) sulfide precipitate forms Write the complete molecular equation for this reaction; Write the ionic equation

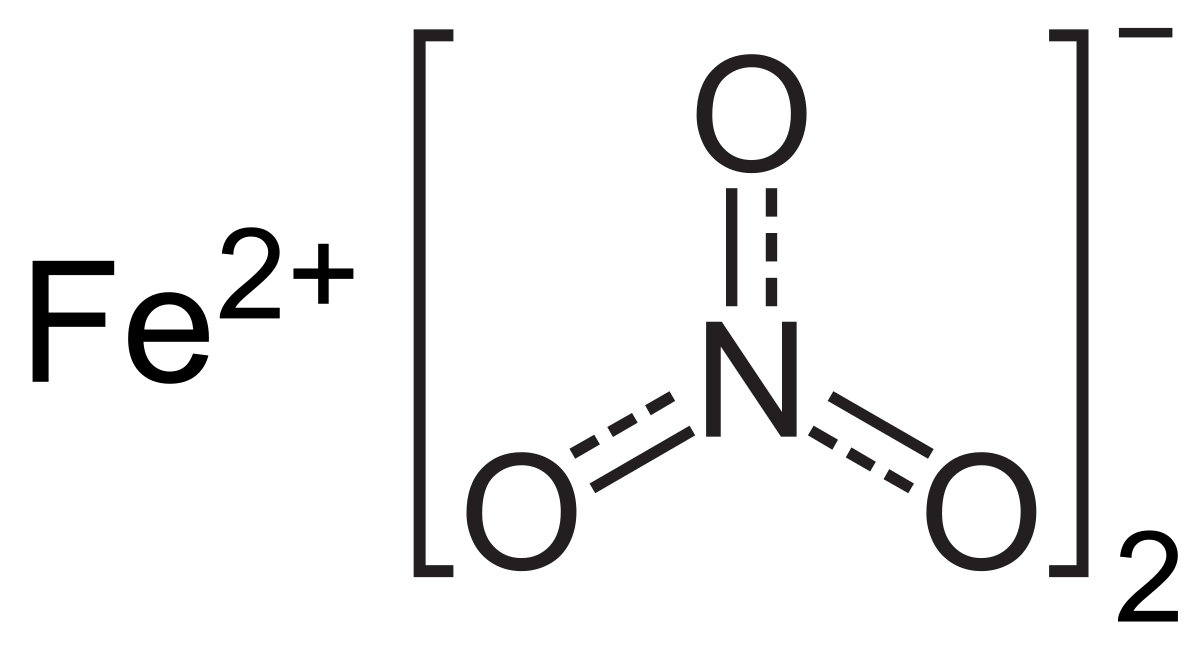








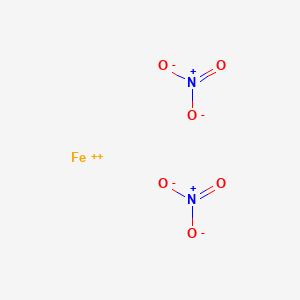







![Solved 17. Iron(II) nitrate [Fe(NO3)2 (aq)] reacts with | Chegg.com Solved 17. Iron(II) nitrate [Fe(NO3)2 (aq)] reacts with | Chegg.com](https://media.cheggcdn.com/media/64b/64b7e906-8e0c-43f2-97f5-f1ae230f5ba3/phpv01T0v.png)