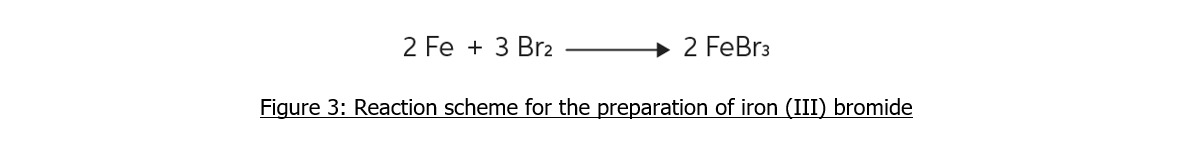
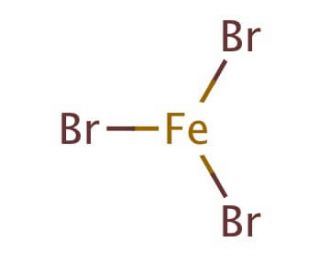
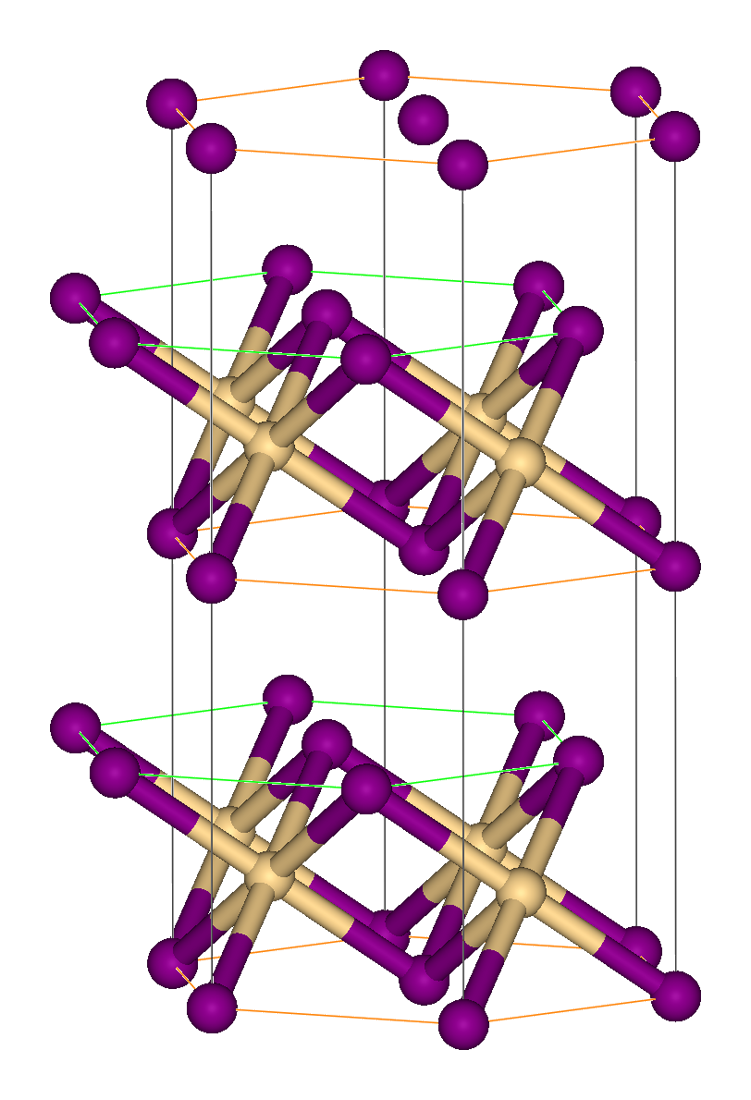
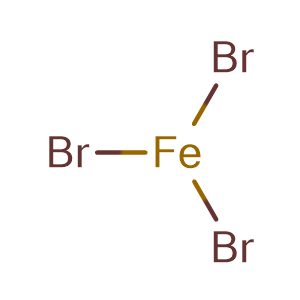
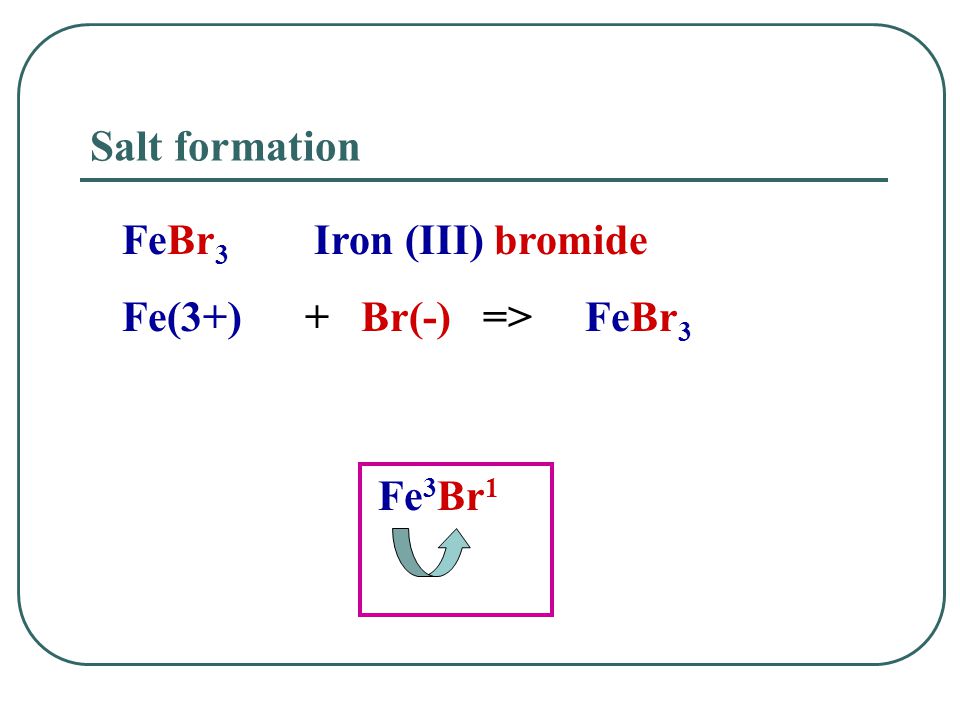
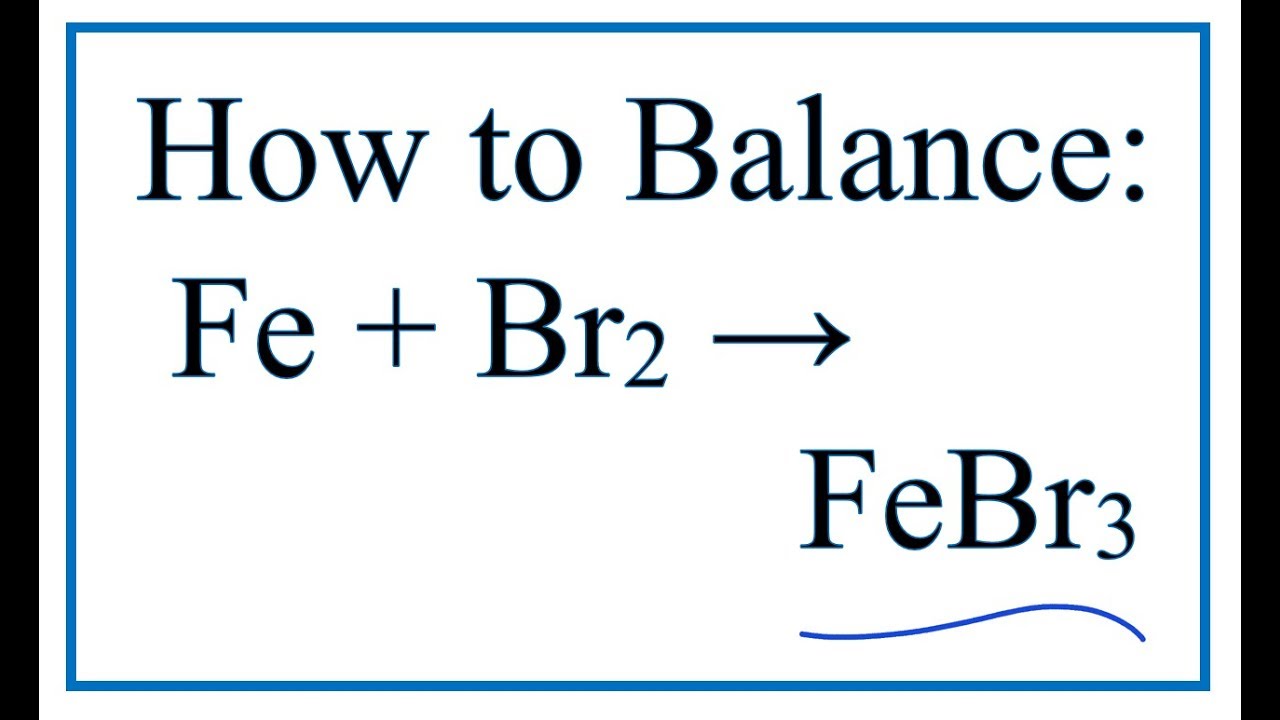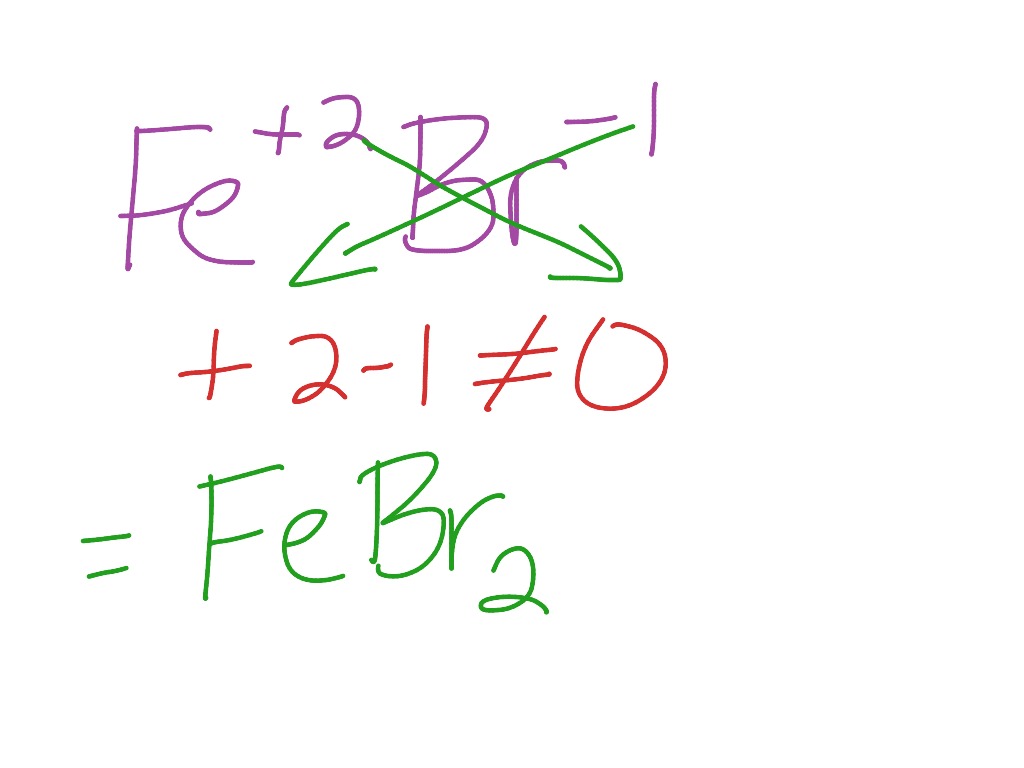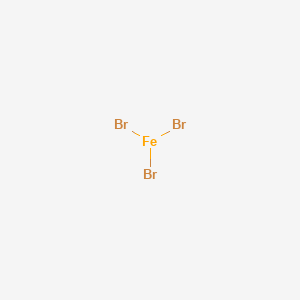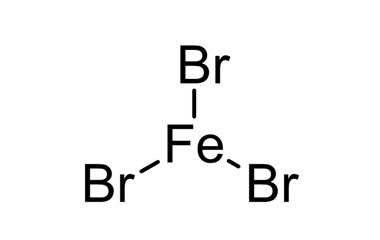Iron(III)Bromide - Iron(III)Bromide,Cupric Salts,Xanthan Gum,CMC,Citric Acid,TCCA,Water Treatment Chemicals Supplier,Food Additives Supplier-Sunvel Chemical Co.,Ltd
Iron (III) bromide and sodium phosphate react to form a precipitate. What is the mass of sodium phosphate required to prepare a 17.1 g precipitate? - Quora

Iron(II) bromide, ultra dry, 99.995% (metals basis), Thermo Scientific Chemicals | Fisher Scientific
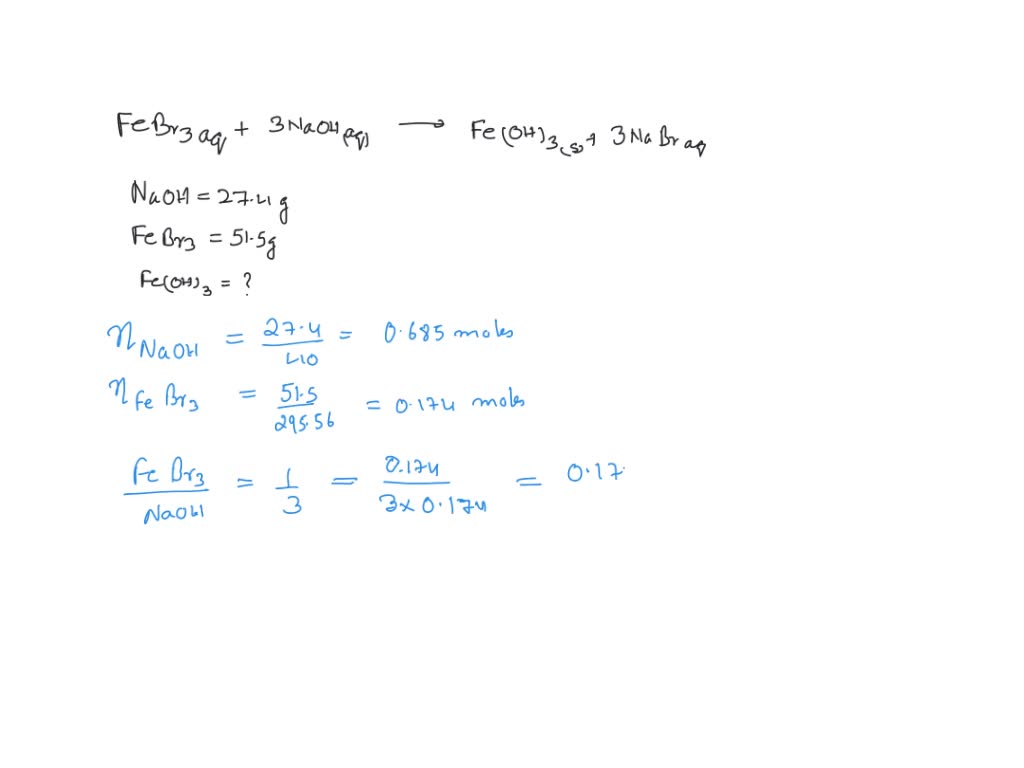

%20Bromide%20(FeBr3).jpg)