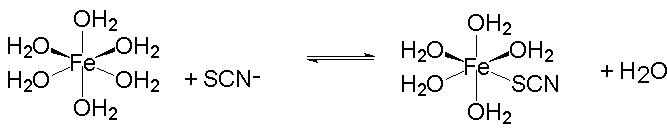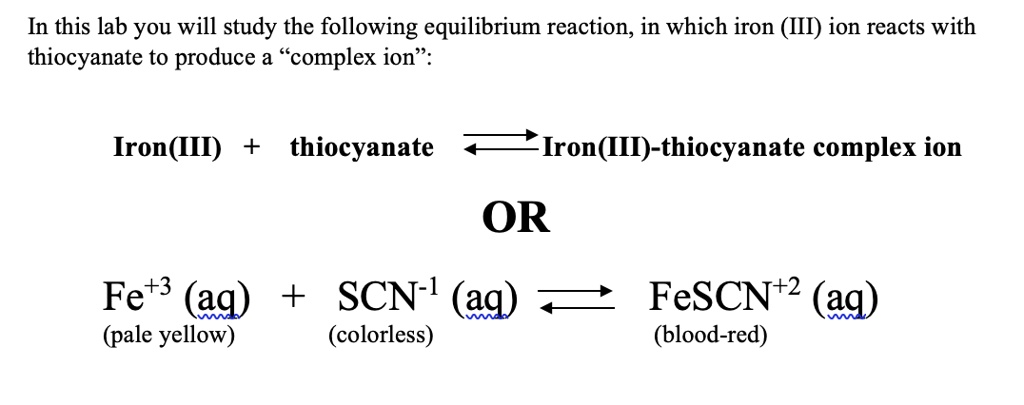
SOLVED: In this lab you will study the following equilibrium reaction; in which iron (III) ion reacts with thiocyanate to produce a "complex ion" Iron (III) thiocyanate Iron(III)-thiocyanate complex ion OR Fe+3 (aq) +

homework - What is the appropriate way of writing the Iron/thiocyanate equilibrium? - Chemistry Stack Exchange
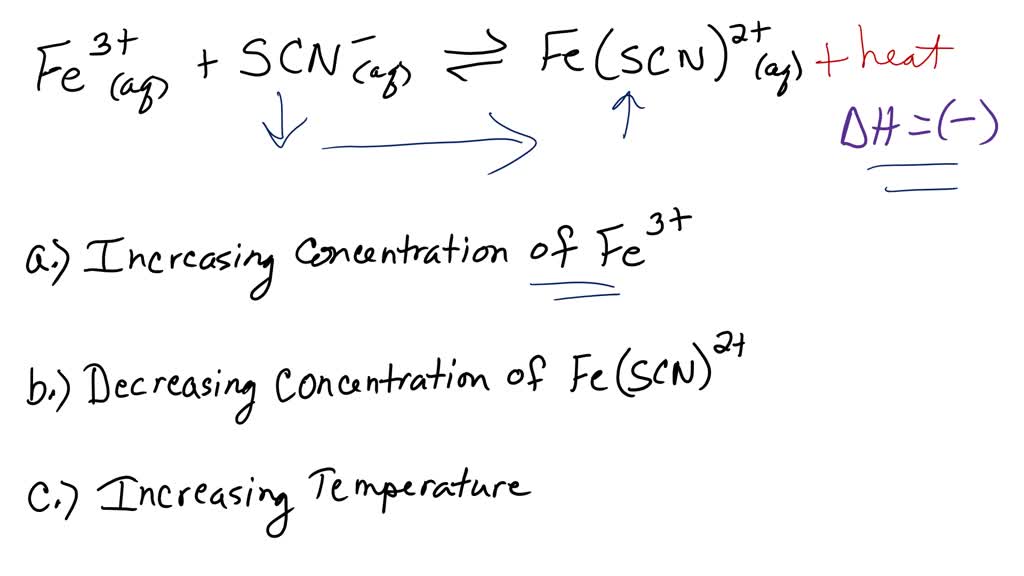
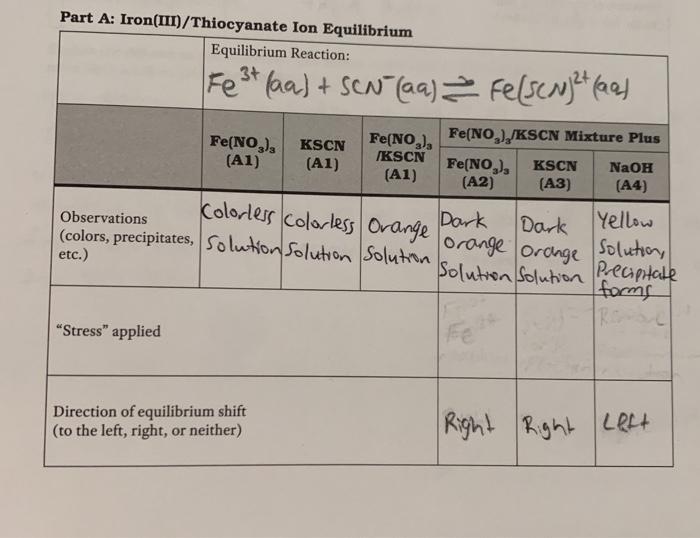
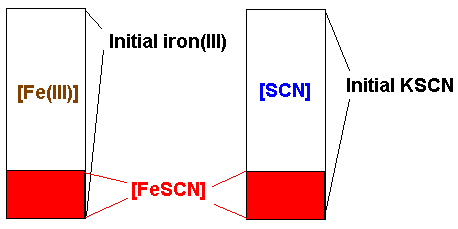
SOLVED: The formation of the iron(III) thiocyanate complex ion is an exothermic equilibrium system. For each change to the system, indicate how the concentration of each species in the chemical equation will
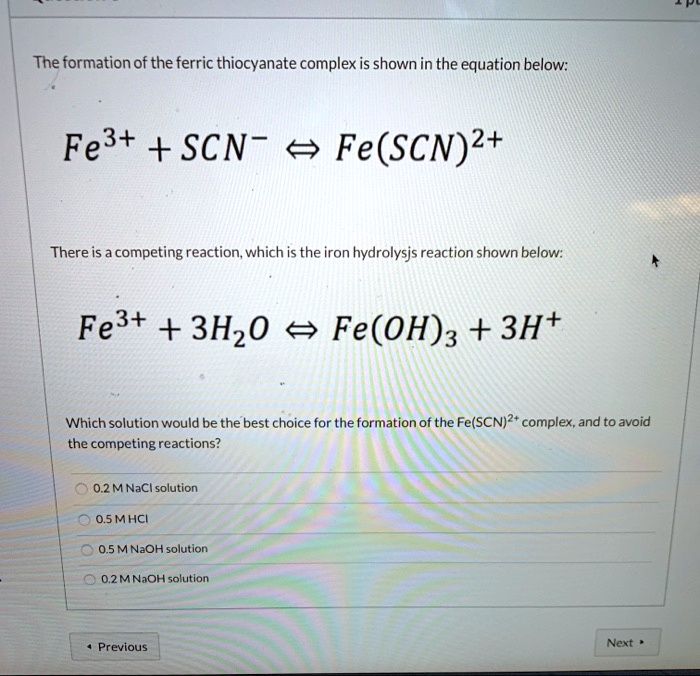
SOLVED: The formation of the ferric thiocyanate complex is shown in the equation below: Fe3+ + SCN- Fe(SCN)+ There is a competing reaction; which is the iron hydrolysjs reaction shown below: Fe3t +

The formation of the iron(III) thiocyanate complex ion is an exothermic equilibrium system. For each change - Brainly.com

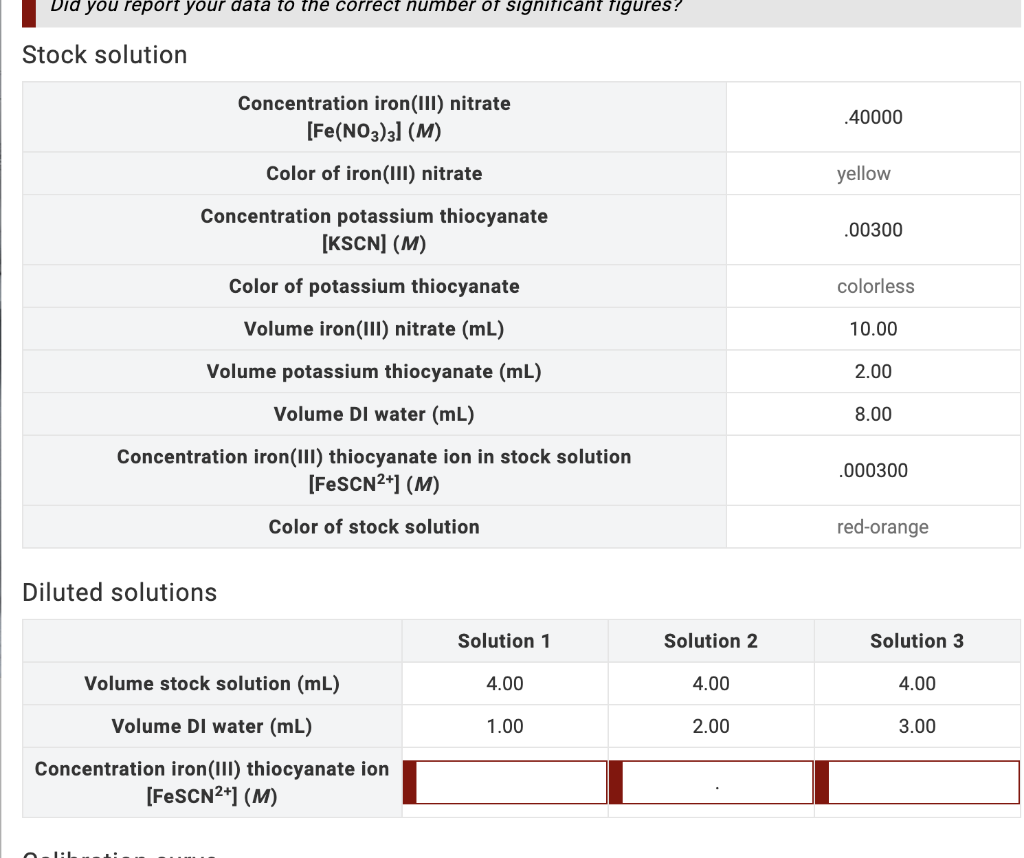
Solved: calculate iron(iii) thiocyanate complex ion concentration (in m) for each solution. record in lab - Brainly.com