A,B) The atmospheric pressure argon plasma jet kINPen and principle of... | Download Scientific Diagram

Group 0/18 NOBLE GASES physical properties uses helium neon argon krpton xenon radon melting points boiling points atomic radii density inertness explained gcse chemistry KS4 science igcse O level revision notes

Noble GasesNoble Gases Helium- Not reactive. Neon- Not reactive. Argon- Not reactive. Krypton- Not reactive. Xenon- Not reactive. Radon- Not. - ppt download
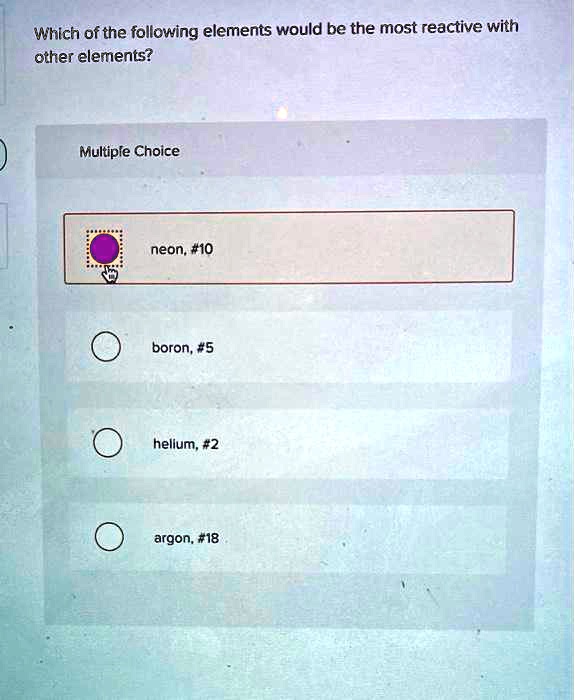
SOLVED: Which of the following elements would be the most reactive with other elements? Multipie Choice neon; #10 boron, #5 argon, #18 hellum;
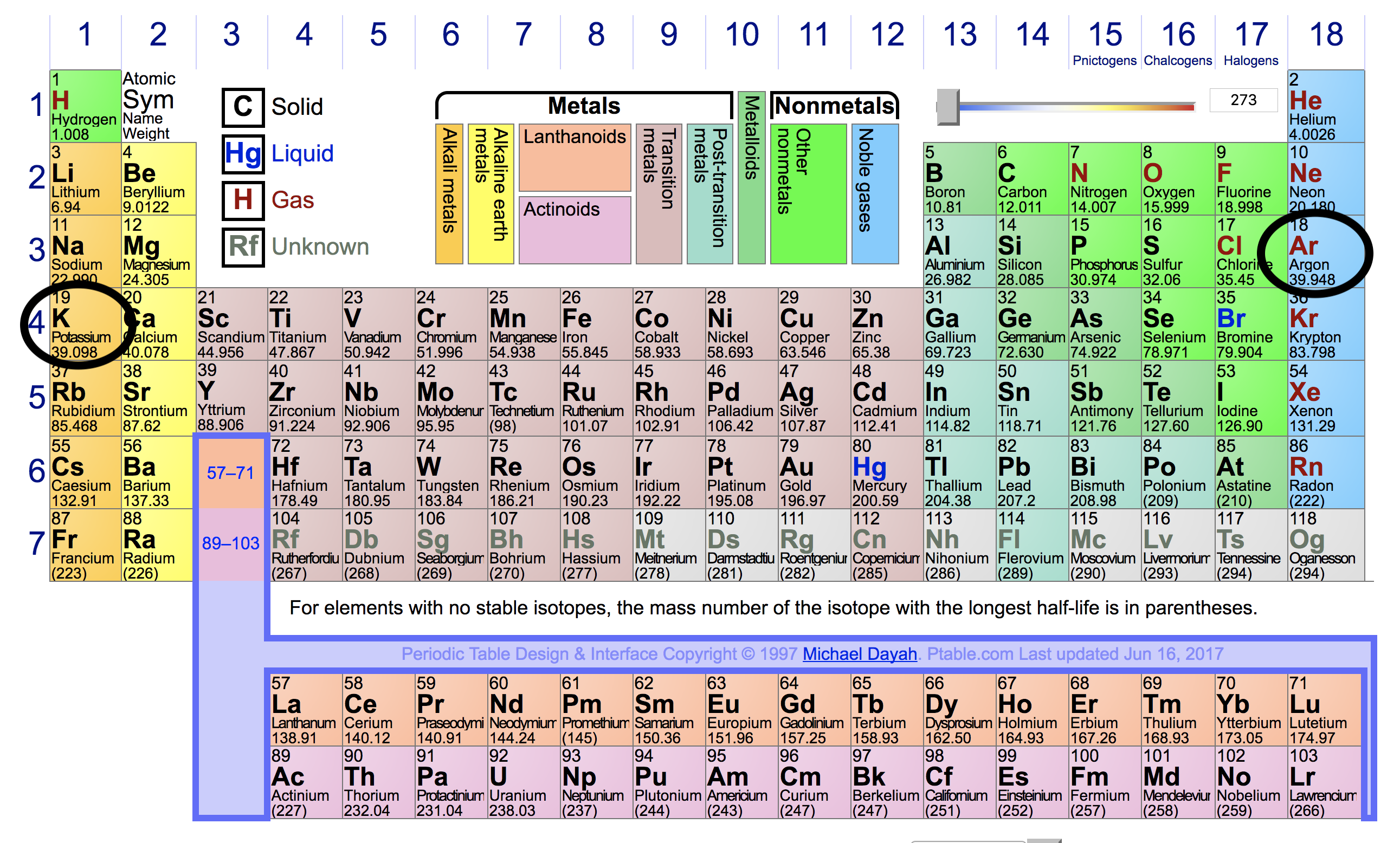
Potassium is highly reactive metal, while argon is an inert gas. How can you explain this difference based on their electron configurations? | Socratic

Noble GasesNoble Gases Helium- Not reactive. Neon- Not reactive. Argon- Not reactive. Krypton- Not reactive. Xenon- Not reactive. Radon- Not. - ppt download


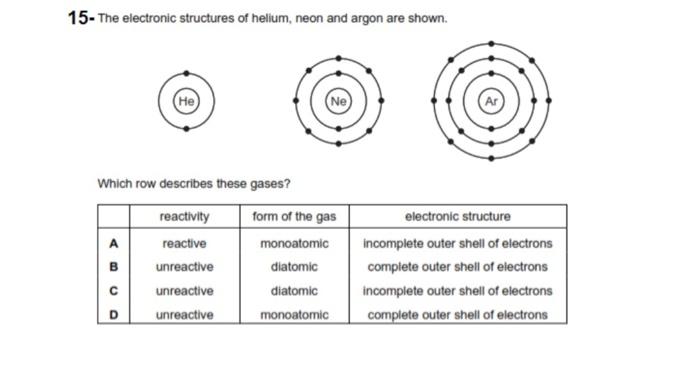








:max_bytes(150000):strip_icc()/argon1-57e1ba9e3df78c9cce33930f.jpg)