Formal oxidation state and calculated charge state from Bader analysis... | Download Scientific Diagram

Which of the following oxidation states are the most characteristics for lead and tin, respectively?

Origin of Sn( ii ) oxidation in tin halide perovskites - Materials Advances (RSC Publishing) DOI:10.1039/D0MA00245C

In view of the signs of ΔrG^o for the following reactions, PbO2 + Pb→ 2PbO, ΔrG^o 0 Which oxidation states are more characteristic for lead and tin?

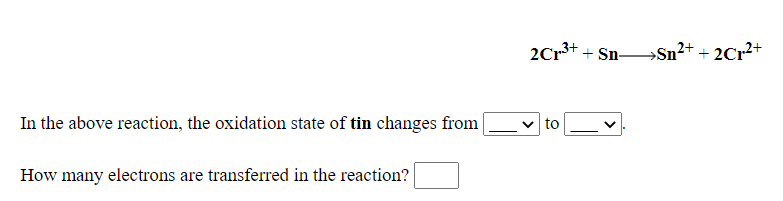
SOLVED: 2Cr(OH)3 3Sn032-+ 4OH" 2Cr042-+ 3Sn0z2-+ SH,O In the above reaction, the oxidation state of tin changes from How many electrons are transferred in the reaction? Submit Answer Retry Entire Group 3

Oxidation states of Cu, Sn, and S in various binary and ternary compounds. | Download Scientific Diagram

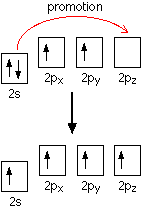
Sec. 7.1 & 9.1: Formation and Naming of Ions Valence Electrons The electrons responsible for the chemical properties of atoms, and are those in the outer. - ppt download

Disproportionation and ligand lability in low oxidation state boryl-tin chemistry | Inorganic Chemistry | ChemRxiv | Cambridge Open Engage