A) Overview of the reactivity of β-O-4 models 1a−d and 2a. R 1 = H for... | Download Scientific Diagram
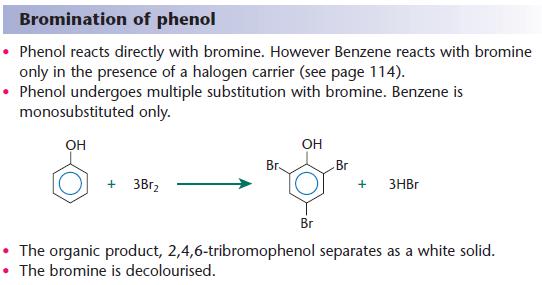
Phenols | a2-level-level-revision, chemistry, aromatics-amines-amino-acids-polymers, phenols | Revision World

Molecules | Free Full-Text | Theoretical Study of the Reaction Mechanism of Phenol–Epoxy Ring-Opening Reaction Using a Latent Hardening Accelerator and a Reactivity Evaluation by Substituents
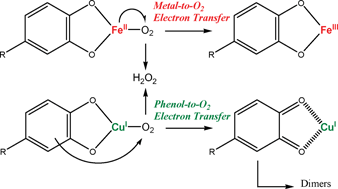
Reactivity of food phenols with iron and copper ions: binding, dioxygen activation and oxidation mechanisms - Food & Function (RSC Publishing)

Number of compounds which are more reactive than phenol towards electrophilic aromatic substitution reaction is:

Phenol Reduces Nitrite to NO at Copper(II): Role of a Proton-Responsive Outer Coordination Sphere in Phenol Oxidation | Journal of the American Chemical Society

Reactivity of a 10-I-3 Hypervalent Iodine Trifluoromethylation Reagent With Phenols | The Journal of Organic Chemistry