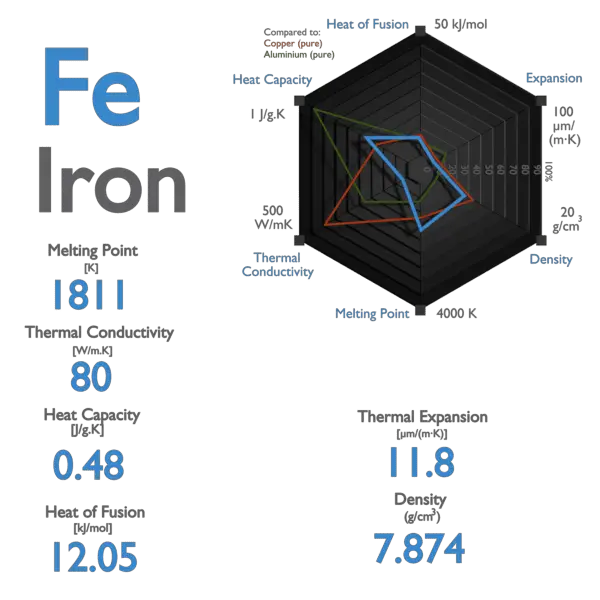
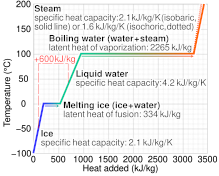
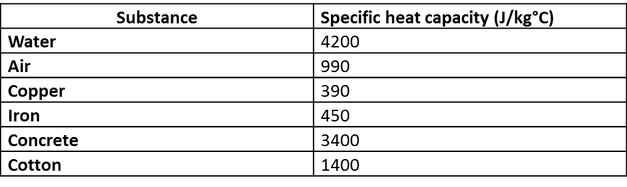
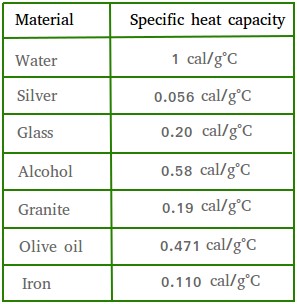
Calculate the amount of heat required to raise the temperature of 5 g of iron from `25^(@)C \"to\" - YouTube
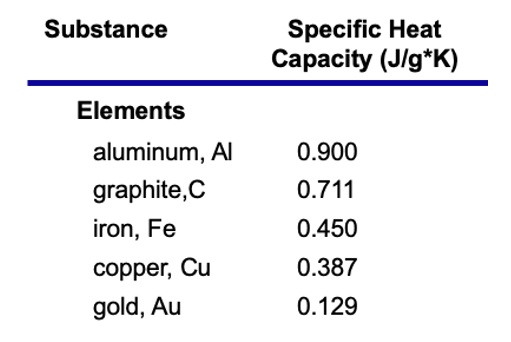
SOLVED: Substance Specific Heat Capacity (Jlg*K) Elements aluminum, Al graphite,C iron, Fe copper; Cu gold, Au 0.900 0.711 0.450 0.387 0.129

Calculate the energy required to heat 790.0g of iron from −2.6°C to 14.9°C. Assume the specific heat - Brainly.com

Comparison between the calculated and experimental heat capacity of... | Download Scientific Diagram
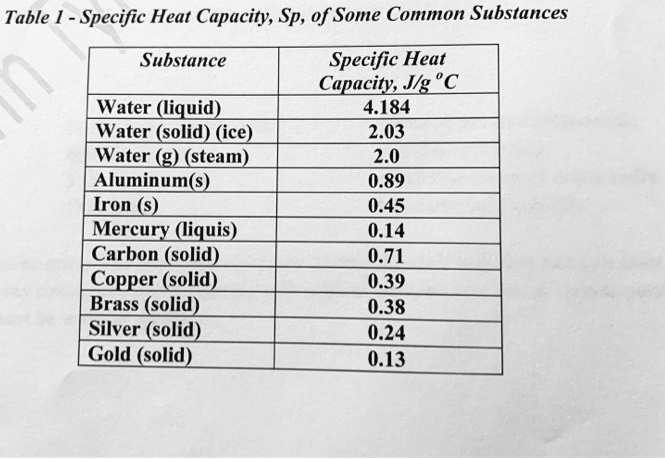
SOLVED: Table 1 Specific Heat Capacity, Sp, of Some Common Substances Substance Specific Heat Capacity, Jg 4.184 2.03 2.0 0.89 0.45 0.14 0.71 0.39 0.38 0.24 0.13 Water (liquid) Water (solid) (ice)
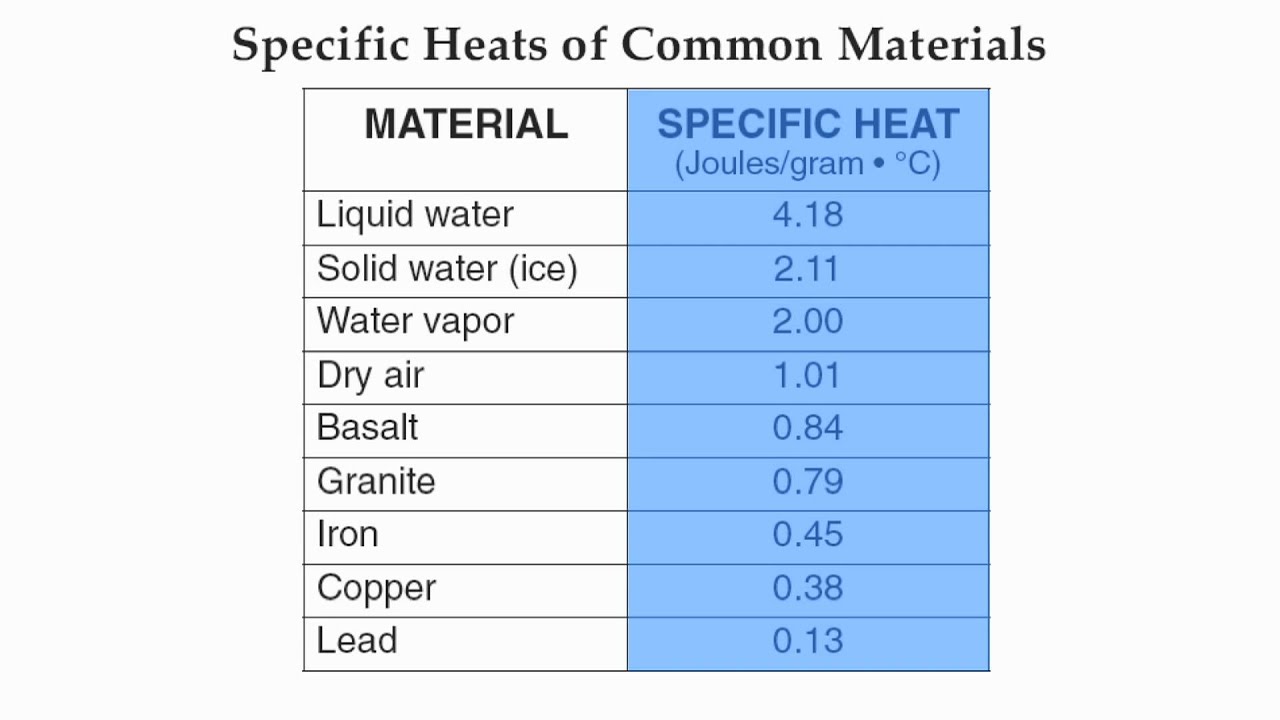
Reference Table Page 1-Specific Heat of Common Materials-Hommocks Earth Science Department - YouTube

Variations of specific heat (plot a) and thermal conductivity (plot b)... | Download Scientific Diagram
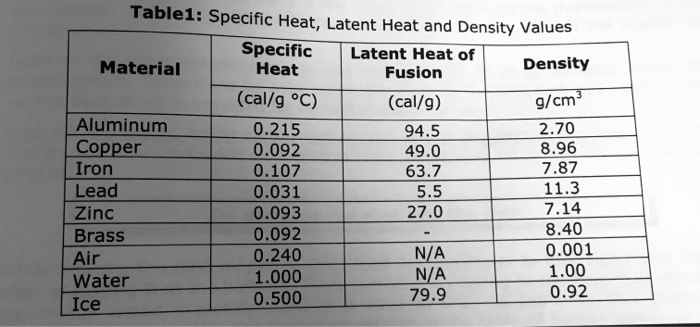
SOLVED: Table1: Specific Heat, Latent Heat ad Density Values Specific Latent Heat of Material Heat Density Fusion (cal/g 'C) (cal/g) g/cm Aluminum 0.215 94.5 2.70 Copper 0.092 49 8.96 Iron 0.107 63
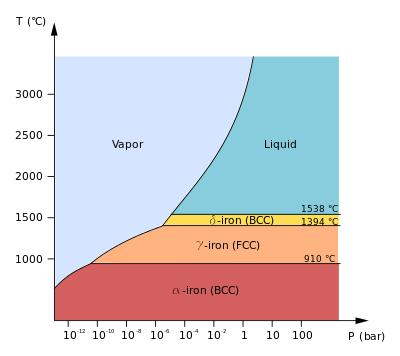
Molten iron is extremely hot, averaging about 1,500 C. The specific heat of iron is 0.46 J/gC. How much heat is released to the atmosphere when 1 kg molten iron cools to
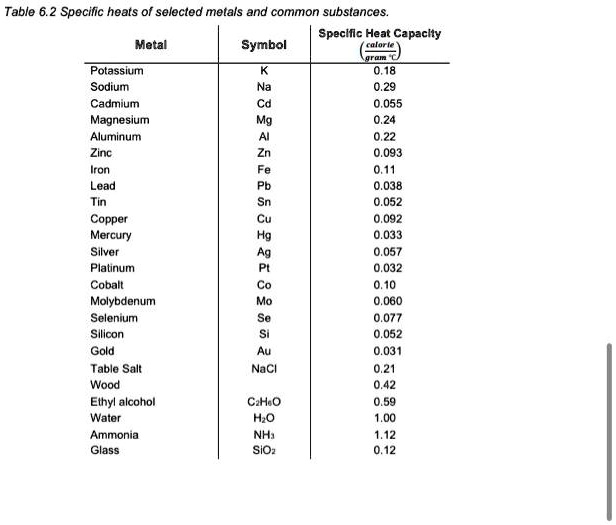
SOLVED: Table 6.2 Specific heats of selected metals and common substances: Speclllc Heat Capacly Metal Symbol Potassium Sodium Cadmium Magnesium Aluminum Zinc Iron Lead 0,18 055 093 0,11 0.038 052 092 033